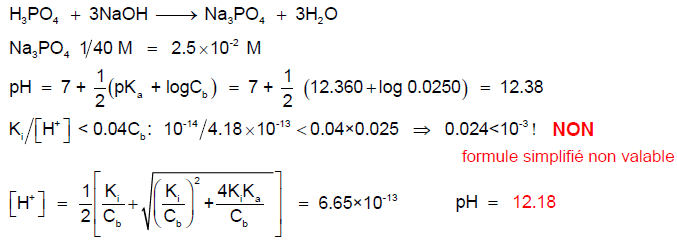![pH of 0.1 M H3PO4 acid solution is :[For the given acid: Ka1 = 10^-3, Ka2 = 10^-7 and Ka3 = 10^-12 ] pH of 0.1 M H3PO4 acid solution is :[For the given acid: Ka1 = 10^-3, Ka2 = 10^-7 and Ka3 = 10^-12 ]](https://haygot.s3.amazonaws.com/questions/1844080_1287766_ans_5cdaae94561f4a2599341308914a04c0.jpg)
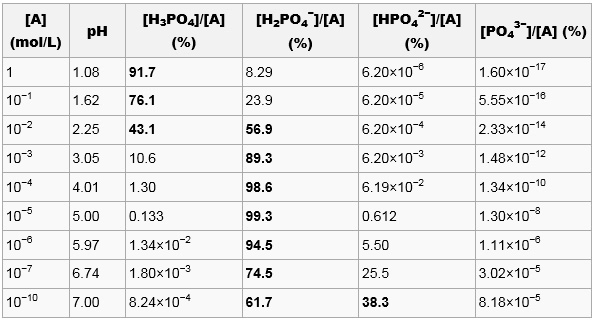
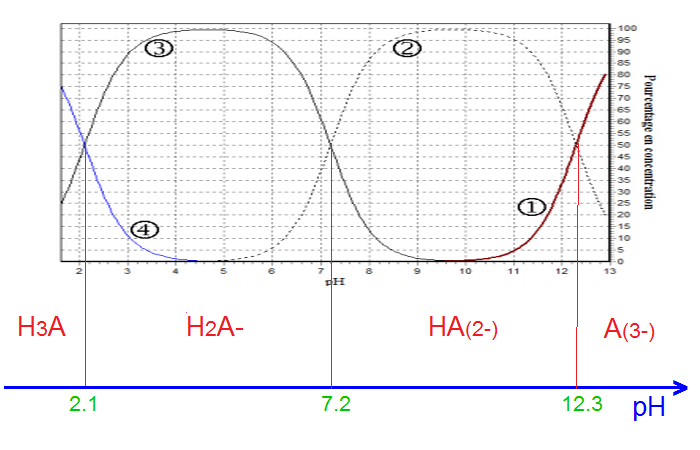
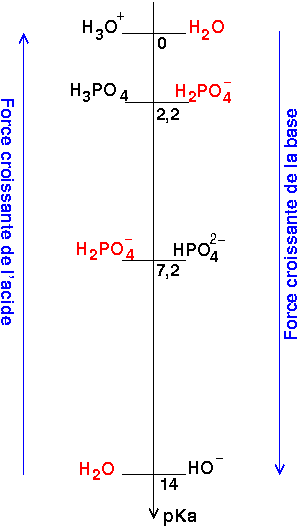
pH of 0.1 M H3PO4 acid solution is :[For the given acid: Ka1 = 10^-3, Ka2 = 10^-7 and Ka3 = 10^-12 ]

Polyprotic Acid Base Equilibria Problems, pH Calculations Given Ka1, Ka2 & Ka3 - Ice Tables - YouTube

The experimental and calculated pH values of phosphoric acid solutions... | Download Scientific Diagram
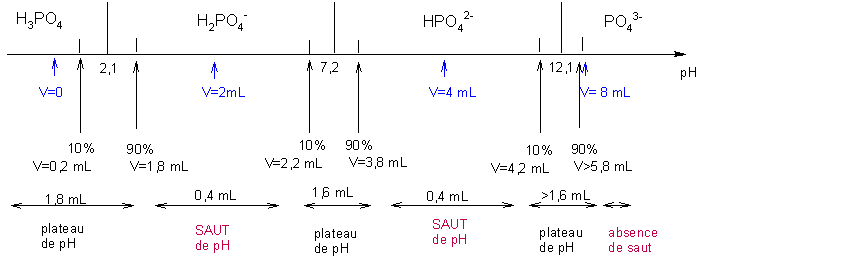
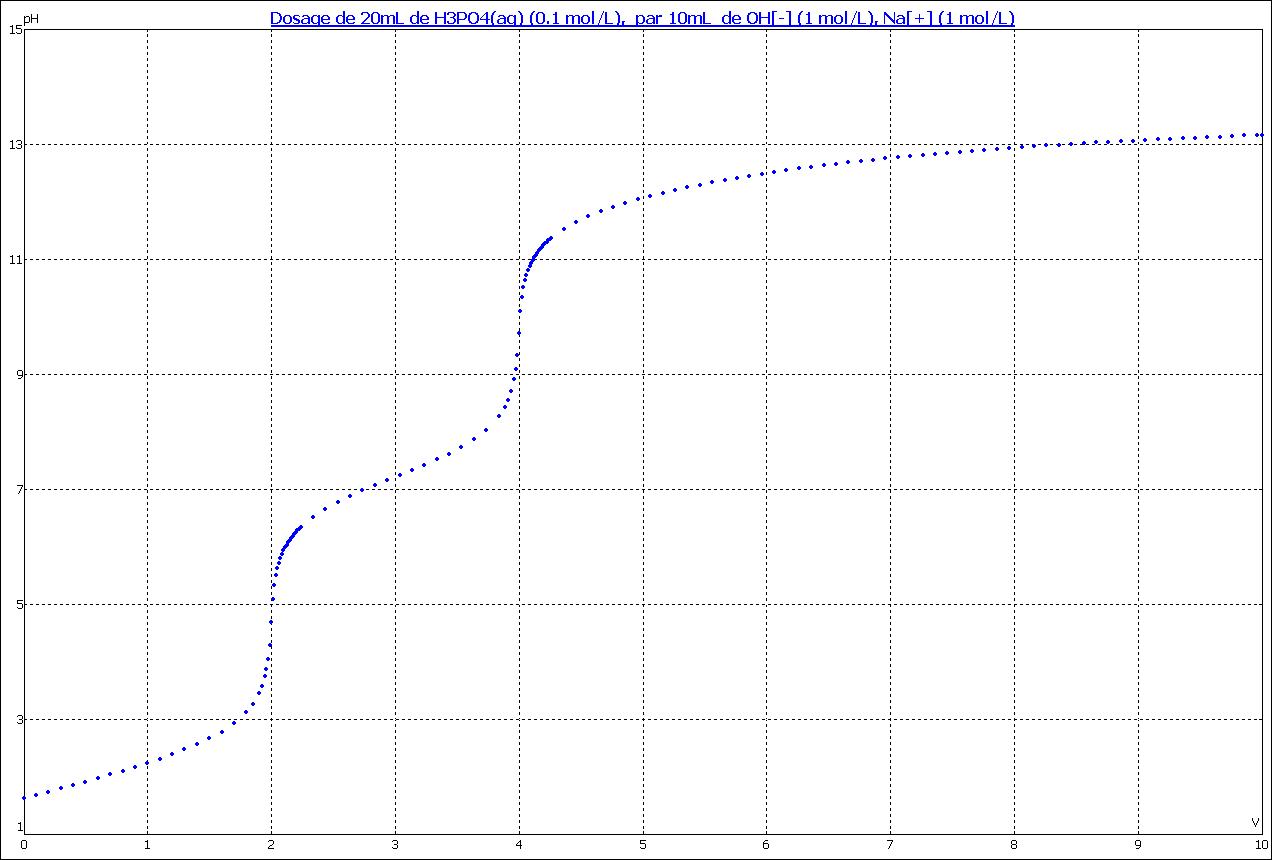
bac CLPI chimie de laboratoire 09/2004 Dosage pHmétrique d'une solution d'acide phosphorique, radioactivité uranium, synthèse organique

Acide orthophosphorique, pur, environ 100 % H3PO4 (contient quelques H4 P2O7), Fisher Chemical | Fisher Scientific




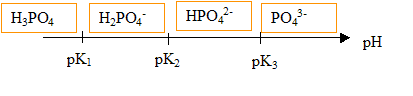
![Change of phosphate species with pH in H3PO4 [5]. | Download Scientific Diagram Change of phosphate species with pH in H3PO4 [5]. | Download Scientific Diagram](https://www.researchgate.net/publication/261995856/figure/fig10/AS:269753218498564@1441325587360/Change-of-phosphate-species-with-pH-in-H3PO4-5.png)